 Kung Flu in Canada is reported at Coronavirus disease (COVID-19): Outbreak update
Kung Flu in Canada is reported at Coronavirus disease (COVID-19): Outbreak update
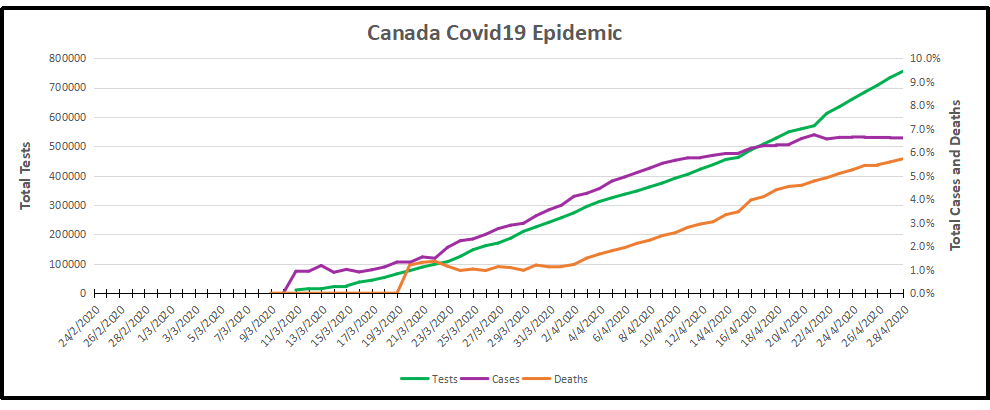
The image presents coronavirus data as of the latest statistics complete to April 28, 2020. Note that Quebec accounts for half of all cases and deaths, most of them in Montreal, and mostly elderly residents in group homes. From the underlying data we can see that this covid 19 outbreak started toward the end of the annual flu season. Here are the daily reported tests, cases, and deaths smoothed with 7 day averaging.
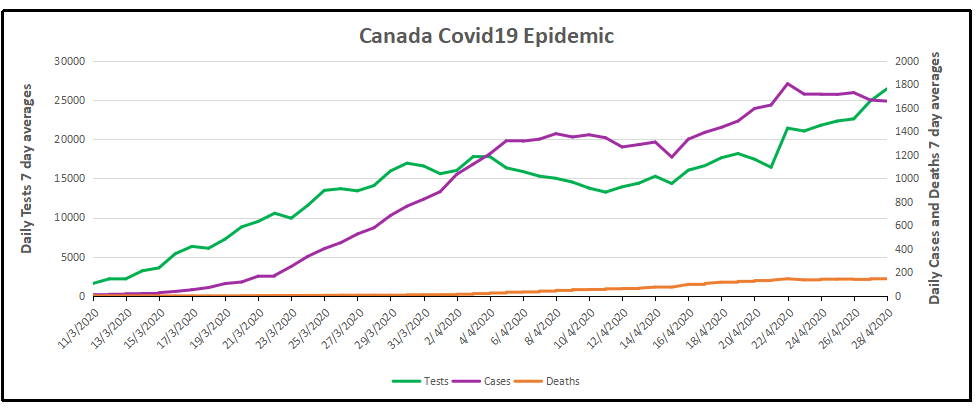 Note how cases early on rose along with the higher daily rates of testing, then held steady April 4 to 15 as testing declined slightly. Both testing and new cases then rose peaking on April 22 with 1806 new cases. Since then testing has ramped up sharply while new cases have tailed off to 1657 yesterday. Deaths per day have been flat at 146 in the last week. (All daily figures are averages of the 7 day period ending with the stated date.) Tests also peaked at 26,417 on April 28, about twice the daily number when April started.
Note how cases early on rose along with the higher daily rates of testing, then held steady April 4 to 15 as testing declined slightly. Both testing and new cases then rose peaking on April 22 with 1806 new cases. Since then testing has ramped up sharply while new cases have tailed off to 1657 yesterday. Deaths per day have been flat at 146 in the last week. (All daily figures are averages of the 7 day period ending with the stated date.) Tests also peaked at 26,417 on April 28, about twice the daily number when April started.
 The cumulative graph shows how the proportions held during this period.
The cumulative graph shows how the proportions held during this period.
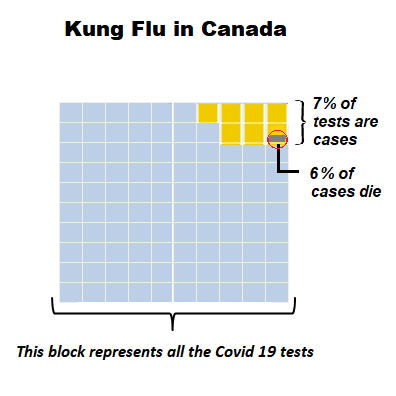
Out of a total 754,800 tests, 50,015 (6.6%) cases were detected, the same rate holding since April 20. 2815 have died (5.7% of cases).
Background from Previous Post
With coronavirus sucking all the air out of room globally, I got interested in looking at how the Canadian national flu seasons compare with the new Wuhan virus. The analysis is important since there are many nations at higher latitudes that are in equilibrium relative to infectious diseases, but vulnerable to outbreaks of new viruses. Where I live in Canada, we have winter outbreaks every year, but are protected by a combination of sanitary practices, health care system and annual vaccines, contributing to herd immunity.
For example, 2018-19 was slightly higher than a typical year, with this pattern:
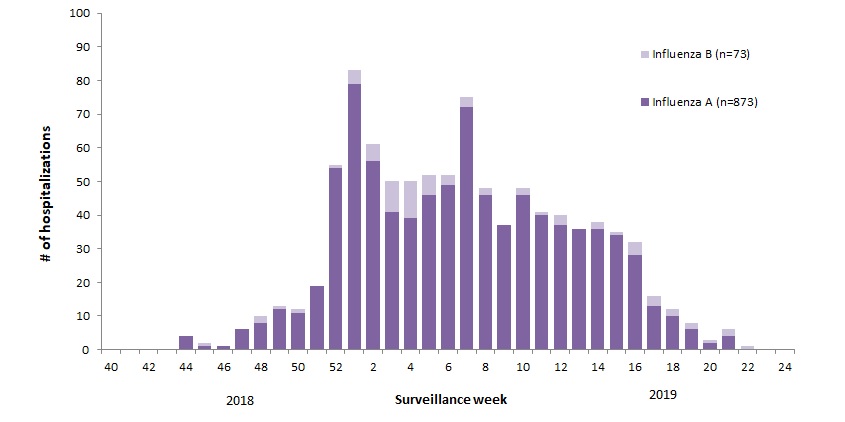
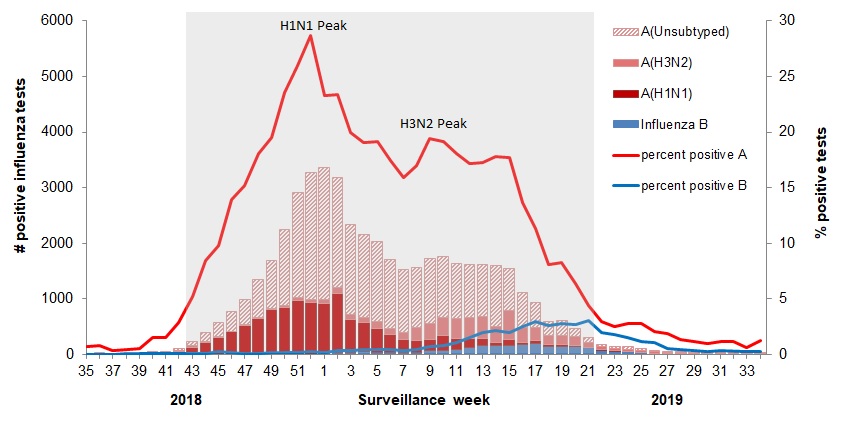 The various flu types are noted, all together making a total of 48,818 influenza detections during the 2018-19 season. A total of 946 hospitalizations were reported by CIRN-SOS sentinels that season (age = or >16). Source: Annual Influenza Reports
The various flu types are noted, all together making a total of 48,818 influenza detections during the 2018-19 season. A total of 946 hospitalizations were reported by CIRN-SOS sentinels that season (age = or >16). Source: Annual Influenza Reports
A total of 137 (14%) ICU admissions and 65 (7%) deaths were reported. The seasonality is obvious, as is the social resilience, when we have the antibodies in place.
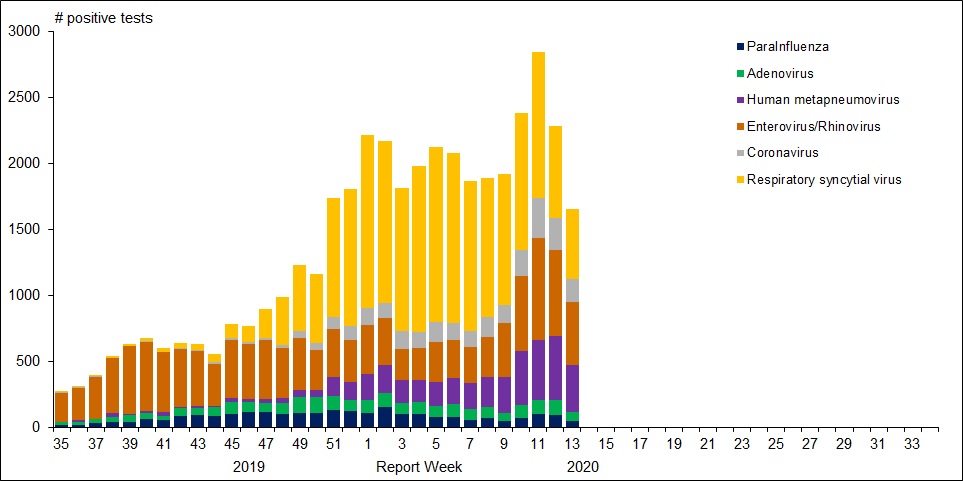
For further background, look at the latest Respiratory Virus Report for week 13 ending March 28, 2020. [In this Respiratory Virus Report, the number of detections of coronavirus reflects only seasonal human coronaviruses, not the novel pandemic coronavirus (SARS-CoV2) that causes COVID-19. Kung Flu statistics are above at the beginning.]
For the period shown in the graph, 320560 flu tests were done, resulting in 32751 type A positives and 22683 type B positives. That is a ratio of 17% of tests confirming conventional flu infection cases. Public Health Canada went on to say in reporting March 22 to 28, 2020 (week 13):
The percentage of tests positive for influenza fell below 5% this week. This suggests that Canada is nearing the end of the 2019-2020 influenza season at the national level. [Keep that 5% in mind]
Summary: It’s true that total cases and deaths are still rising, and everyone should practice sanitary behaviors and social distancing. But it appears that we are weathering this storm and have the resources to beat it. Let us hope for reasonable governance, Spring weather and a return to economic normalcy.
Postscript: Good News from Calgary and Hamilton
The Calgary Herald reports U of C researchers to begin hydroxychloroquine trial on COVID-19 patients. Excerpts in italics with my bold.
A provincewide clinical trial led by the University of Calgary will test the effectiveness of the anti-malarial drug hydroxychloroquine on COVID-19 patients, with the goal of reducing pressure on hospitals and preventing further infections.
“There is minimal evidence for use of hydroxychloroquine to use it, but there is enough (evidence) to study it,” said Metz, the acting facility medical director at Foothills Medical Centre and a professor in the department of clinical neurosciences at the Cumming School of Medicine.
“It just has to be done. If this drug does, indeed, reduce the severity and help people get better faster, it can help us in flattening the curve.”
The “HOPE” trial, to begin April 15, will target 1,600 Alberta outpatients who have tested positive for COVID-19 and are at risk of developing severe symptoms. The study will determine if hydroxychloroquine can prevent hospitalization for those at highest risk of developing a severe illness.
Participants will give their permission to Alberta Health Services after testing positive for COVID-19 and provide their contact information to U of C researchers. They’ll then be screened for safety and eligibility through a telephone interview and review of their electronic health record.
Those patients accepted will be sent hydroxychloroquine to their homes and will be required to take the drug over a five-day period. Researchers will follow up with participants seven and 30 days after starting the treatment.
Metz said timing of the trial is crucial and must begin within 96 hours of confirmation of a positive COVID-19 result, and within 12 days of symptom onset.
Should the drug prove effective, it may reduce the pressures that COVID-19 is expected to put on the health-care system.
“If we can keep more people out of hospital, then we’re not going to have that huge rise in hospitalizations and more people can get better at home,” she said. “Our system will get back to normal life or whatever we choose to move to in the future.
“We’ll be able to get there if we find this works.”
Hamilton Health Services starting an Anti-Covid19 study this month
Title: Anti-Coronavirus Therapies to Prevent Progression of COVID-19, a Randomized Trial
The ACT COVID-19 program consists of two parallel trials evaluating azithromycin and chloroquine therapy (ACT) versus usual care in outpatients and inpatients who have tested positive for COVID-19. The trial is an open-label, parallel group, randomized controlled trial with an adaptive design. Adaptive design features include adaptive intervention arms and adaptive sample size based on new and emerging data.
Experimental: Azithromycin and Chloroquine Therapy (ACT)
Chloroquine (Adults with a bodyweight ≥ 50 kg: 500 mg twice daily for 7 days; Adults with a bodyweight < 50 kg: 500 mg twice daily on days 1 and 2, followed by 500 mg once daily for days 3-7), plus Azithromycin (500 mg on day 1 followed by 250 mg daily for 4 days) Source: ClinicalTrials.gov
See also Preemptive Coronavirus Therapy Positive Update
Some coronavirus humor.