Water (H2O) has magical properties that make our planet suitable for us. The video explains why most of the ocean water is about 4 degrees Celsius. A transcript from another presentation draws the implications. Excerpts in italics with my bolds.
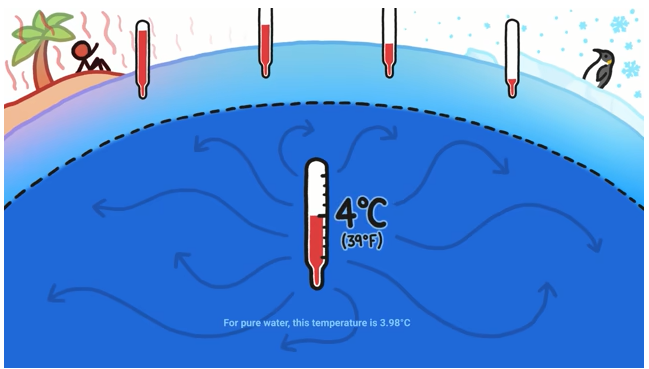
At the surface, ocean water can vary wildly in temperature – the water at the equator is around 30 degrees Celsius and the water at the poles is, well, freezing. But surface waters are only a small fraction of the total water in the ocean. Dive a little deeper, and you’ll find that a whopping 75 percent of the ocean’s water is all at the same temperature…and we’re not talking averages or anything – the vast majority of ocean water is 4 degrees Celsius. And that’s not just a coincidence – it’s because water is weird.
As a liquid cools, its molecules slow down and the liquid generally gets denser and denser. That’s how molten metals, wax, nacho cheese, and basically everything else behaves – except water. Water does become denser as it cools though, but only up to a point. Then it reverses course and actually gets less dense.
This happens because once water molecules slow down enough, intermolecular forces due to the water molecule’s unique shape start pushing the molecules apart until – at zero degrees and below – they form a lattice-like structure. That’s why ice is less dense than water.
But the magic temperature where water is actually densest is 4 degrees Celsius. This weird maximum density is what causes the vast majority of the ocean to be stuck at the same temperature.
By about 1000 meters down, water has cooled to around four degrees. Any water here, or below, that happens to warm up – say, via heat from a hydrothermal vent or underwater volcano – will get a little less dense and float upwards, as less-dense things tend to do – out of this 4-degree zone. Strangely, water cooler than 4 degrees will behave the same way; any water that loses a little bit of heat will also become a little less dense and balloon upwards.
As a result, all the ocean water below 1000 meters or so is about 4 degrees.
Well, almost all the water.
The very deepest parts of the ocean can get just a tiny bit colder, because of salt. When salt ions are stuck to water molecules, they weigh them down, making saltier water a little denser than less salty water. So when polar ice forms, salt gets pushed into the surrounding water, making it super-salty. This super-salty water is most dense slightly below 4 degrees, in addition to being a little denser than less salty water, so, it has the tendency to plummet straight to the seafloor.
This heavier, colder water makes the deepest depths of the ocean slightly colder and denser than the water above. Expeditions to the deepest parts of the ocean, like the Challenger Deep of Mariana’s Trench have recorded temperatures of 1 degree. However, the same rules apply down there as they do in the rest of the water column – any water that warms or cools, even a bit, will become less dense and float away into the higher, less dense layers above.
If these weird water density rules didn’t apply – if water behaved like, say, nacho cheese – ocean water would just solidify from the bottom up as it’s cooled, and we wouldn’t have liquid oceans at all.

Perfect illustration of the polar to equator current upwellings. So very interesting.
LikeLike
The whole premise is wrong. My estimate of the average ocean global temperature is +2.
If and/or as the ocean’s warm, they will become more dense. This results in a contraction which will nicely offset any increase in sea level due to melting ice.
LikeLike
Keith, don’t see how your number is possible.
LikeLike
Mine is an estimate cobbled together from various sources I’ll give you that. Your graph seems wrong. By the look of it, the average surface temperature is about 22 degrees. This seems unlikely.
LikeLike
Not my graph Keith. Comes from here:
https://www.windows2universe.org/earth/Water/temp.html
They say the surface (apparently down to 400 meters) averages 17 C. Other websites say measurements below 5500 meters are sparce and unreliable.
LikeLike
Nice science post! Thanks! VR
LikeLike
I am curious, with the very, very high pressure at the bottom of the ocean, wouldn’t that shift the density temperature graph one way or another? My expectation is the graph will move to lower temperature for max density, so adding that to the effect of salt could explain the 3 degree change. Just thinking out loud 😉
LikeLike
Juan, my curiosity led me to see that it’s complicated. There’s not only stratification by temperature and salinity, but lots of dynamics with ocean currents and wind patterns, etc. Here’s what I learned:
LikeLike