 A year ago it was already evident that Ivermectin provided superior protection against SARS CV2 infection compared to Pfizer and Moderna mRNA vaccines. Bruce Sanders sent me the following analysis in pdf.
A year ago it was already evident that Ivermectin provided superior protection against SARS CV2 infection compared to Pfizer and Moderna mRNA vaccines. Bruce Sanders sent me the following analysis in pdf.
Improving Covid 19 Outcomes: A Comparison of Prophylactic Measures
• Ivermectin (IVM) for humans is safe and improves the health of those that contract SARS CoV 2
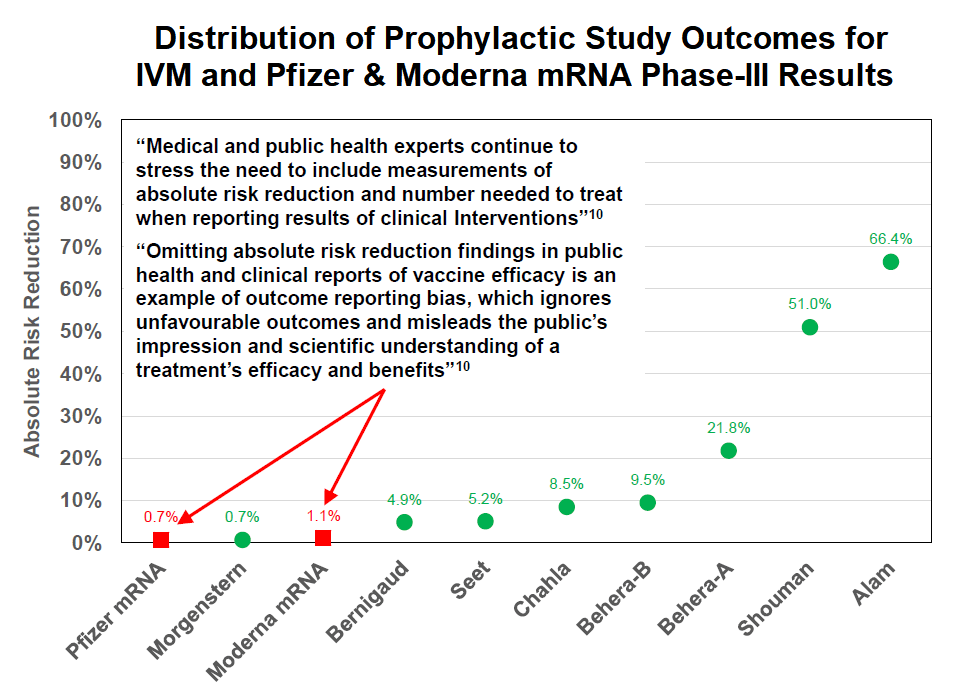
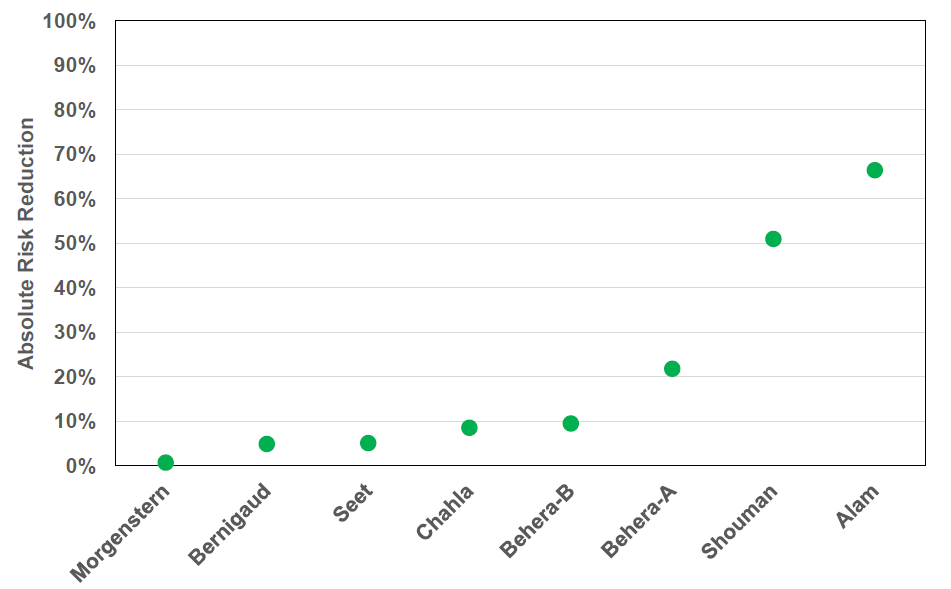
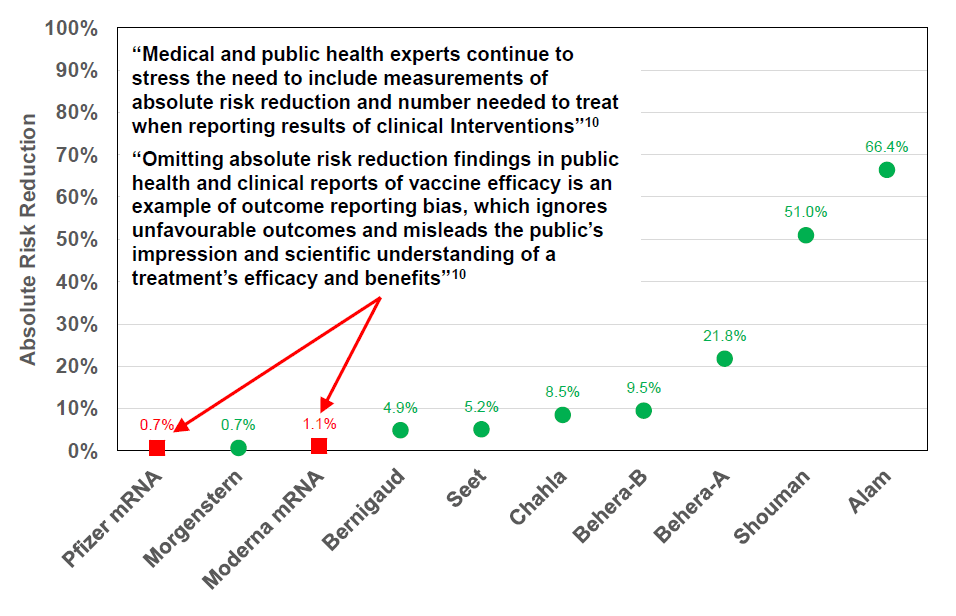
• Absolute Risk Reduction for Ivermectin when used as a prophylactic measure varies from 0.7% to 66%.
• Pfizer and Moderna messenger RNA (mRNA) injections, which are also prophylactic measures, have a 0.7% and 1.1% absolute risk reduction respectively.
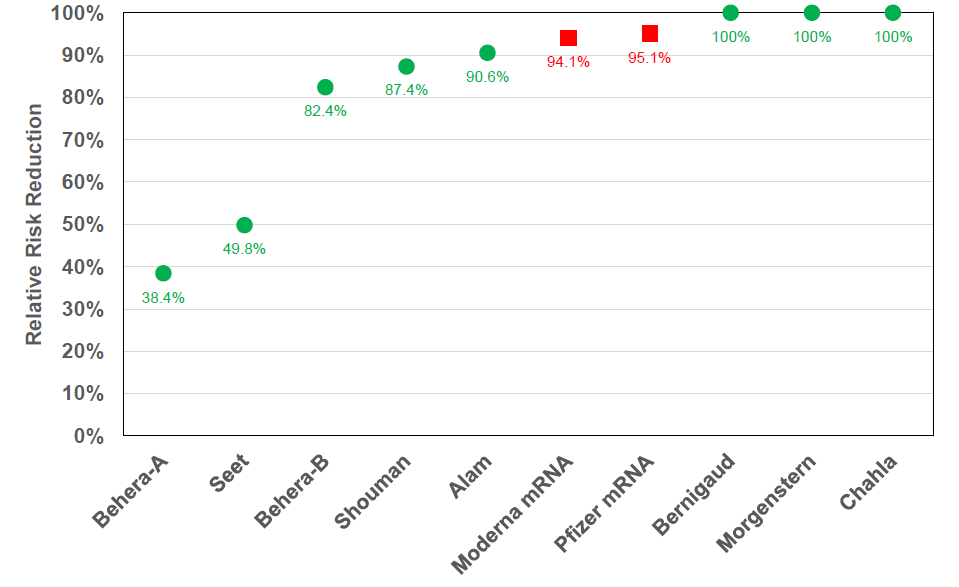
• Relative Risk Reduction for Ivermectin when used as a prophylactic measure varies from 38% to 100%.
• Pfizer and Moderna mRNA injections, which are also prophylactic measures, have a 95% and 94% relative risk reduction respectively.
IVM Studies
• The basis for this work is taken from the ivermectin meta analysis database. 1 Eight of those studies that passed the exclusion assessment are included.
• The risk reduction across the studies is shown here:
Distribution of IVM Prophylactic Study Outcomes
Absolute Risk Reduction
Relative Risk Reduction
Distribution of Prophylactic Study Outcomes for
IVM and Pfizer & Moderna mRNA Phase III Results
Absolute Risk Reduction
Relative Risk Reduction
Discussion –Absolute & Relative Risk Reduction
• Absolute Risk Reduction illustrates the absolute magnitude of disease risk between the treatment and control groups.
• This is the actual difference treatment provides
• Relative Risk Reduction is the reduced risk from treatment relative to the risk in untreated individuals.
• This is a relative difference treatment provides, and says nothing of the magnitude.
• Both are important to understand in the context of making high quality decisions for treatment.
• Consider a case where a person exceeds the road speed limit by 40%.
• A relative 40% excess appears to be significant.
• If the posted speed limit is 20 mph, the absolute excess is only 8 mph
• The extremely low absolute risk reduction of the Pfizer & Moderna mRNA injections does not warrant prohibition of other prophylactic options or a necessity to mandate these injections.
IVM Improves all Outcomes and is Safe for Humans
- “Statistically significant improvements are seen for mortality, hospitalization, recovery, cases, and viral clearance.”1
- “Moderate-certainty evidence finds that large reductions in COVID-19 deaths are possible using ivermectin. Using ivermectin early in the clinical course may reduce numbers progressing to severe disease. The apparent safety and low cost suggest that ivermectin is likely to have a significant impact on the SARS-CoV-2 pandemic globally.”11
- 2010: “Doses as high as 800 micrograms per kilogram were tolerated”12
- 2013: “it (Ivermectin) could be repurposed relatively quickly given that the ivermectin is very safe and already given in mass drug administrations (MDA) to humans around the world”13
- 2015: “We conclude that IVM can be safely given (at 200 µg/kg)”14
- 2018: “Ivermectin at both doses (600 μg/kg and 300 g/kg per day) assessed was well tolerated”15
IVM is Part of Covid-19 Treatment Guides
- Among many others around the world, both the American Association of Physicians & Surgeons (AAPS) Treatment Guide and the Frontline Covid19 Critical Care (FLCCC) Alliance protocols contain proven treatments including prophylactic measures.
- For additional information on the AAPS and FLCCC treatments, consult the following:
- https://aapsonline.org/covidpatientguide/
- https://covid19criticalcare.com/covid-19-protocols/
*** Always consult your physician before taking any medication ***
References
2.Bernigaudet al., Annals of Dermatology and Venereology, doi:10.1016/j.annder.2020.09.231 Ivermectin benefit: from scabies to COVID-19, an example of serendipity.
3.Alamet al., European Journal ofMedicaland Health Sciences, doi:10.24018/ejmed.2020.2.6.599 Ivermectin as Pre-exposure Prophylaxis for COVID-19 among Healthcare Providers in a Selected Tertiary Hospital in Dhaka –An Observational Study.
4.Seetet al., International Journal of Infectious Diseases, doi:10.1016/j.ijid.2021.04.035 Positive impact of oral hydroxychloroquine and povidone-iodine throat spray for COVID-19 prophylaxis: an open-label randomized trial.
5.Behera et al., PLoSONE, doi:10.1371/journal.pone.0247163 Role of ivermectin in the prevention of SARS-CoV-2 infection among healthcare workers in India: A matched case-control study.
6.Shoumanet al., Journal of Clinical and Diagnostic Research, doi:10.7860/JCDR/2020/46795.0000 Use of Ivermectin as a Potential Chemoprophylaxis for COVID-19 in Egypt: A Randomised Clinical Trial.
7.Morgenstern et al., Cureus, doi:10.7759/cureus.17455 Ivermectin as a SARS-CoV-2 Pre-Exposure Prophylaxis Method in Healthcare Workers: A Propensity Score-Matched Retrospective Cohort Study.
8.Behera et al., Cureus13:8, doi:10.7759/cureus.16897 Prophylactic Role of Ivermectin in Severe Acute Respiratory Syndrome Coronavirus 2 Infection Among Healthcare Workers.
9.Chahlaet al., American Journal of Therapeutics, doi:10.1097/MJT.0000000000001433 A randomized trial -intensive treatment based in ivermectin and iota-carrageenan as pre-exposure prophylaxis for COVID-19 in healthcare agents.
10.Brown . Outcome Reporting Bias in COVID-19 mRNA Vaccine Clinical Trials. Medicina(Kaunas). 2021 Feb 26;57(3):199.
11.Bryant et al Ivermectin for Prevention and Treatment of COVID-19 Infection: A Systematic Review, Meta-analysis, and Trial Sequential Analysis to Inform Clinical Guidelines, Am. J. Ther.: July/August 2021 -Volume 28 -Issue 4 -p e434-e460
12.Rea et al. Ivermectin and River Blindness: Science and philanthropy put an end to blindly following the next generation. American Scientist 98 (4), 294–303. 6
13.Syllaet al(Bill & Melinda) Gates Grand Challenges Explorations award: Endectocides for Controlling Transmission of Mosquito-borne Diseases. MalariaworldJ. 2013 Mar;4(5):
14.OuédraogoAL et alEfficacy and safety of the mosquitocidaldrug ivermectin to prevent malaria transmission after treatment: a double-blind, randomized, clinical trial. Clin Infect Dis. 2015 Feb 1;60(3):357-65. Epub2014 Nov 19. Erratum in: Clin Infect Dis. 2016 Sep 1;63(5):715.
- Smit MR et alSafety and mosquitocidalefficacy of high-dose ivermectin when co-administered with dihydroartemisinin-piperaquine in Kenyan adults with uncomplicated malaria (IVERMAL): a randomised, double-blind, placebo-controlled trial. Lancet Infect Dis. 2018 Jun;18(6):615-626. Epub2018 Mar 27.
Other
Elliott, M.H. et al Characteristics and Reporting of Number Needed to Treat, Number Needed to Harm, and Absolute Risk Reduction in Controlled Clinical Trials, 2001–2019. JAMA Intern. Med. 2020, 181.
Thomas, E.T. & Heneghan, C. Outcome Reporting Bias. https://catalogofbias.org/biases/outcome-reportingbias/
Footnote: Clarification on the Math
Absolute Infection Risk Reduction answers the question: What is the difference between the infection rates of the test group and the control group? It is simply subtracting the test group rate from the control group rate.
Relative Infection Risk Reduction answers the question: What percentage of control group infections would be prevented if that group had the same infection rate as the test group? Multiplying the control group population by the test group rate gives a lower number of infections than actually occurred. The relative reduction is the difference as a percentage of the total reported infections in the control group.
So, in the case of the Pfizer vax trial, the test group numbered 21,720 with 8 infections compared to the control group of 21,728 with 162 infections. So the absolute risk reduction was 0.746% less 0.039% equaling 0.7%. The relative risk reduction was (162 – 8) divided by 162 equaling 95%. The relative reduction was impressively large, but it was a reduction upon a very, very small infection rate.
Source of Pfizer data: SARS-CoV-2 Vaccination — An Ounce (Actually, Much Less) of Prevention


Great Content! Thanks for posting!
LikeLike
Reblogged this on HiFast News Feed.
LikeLike